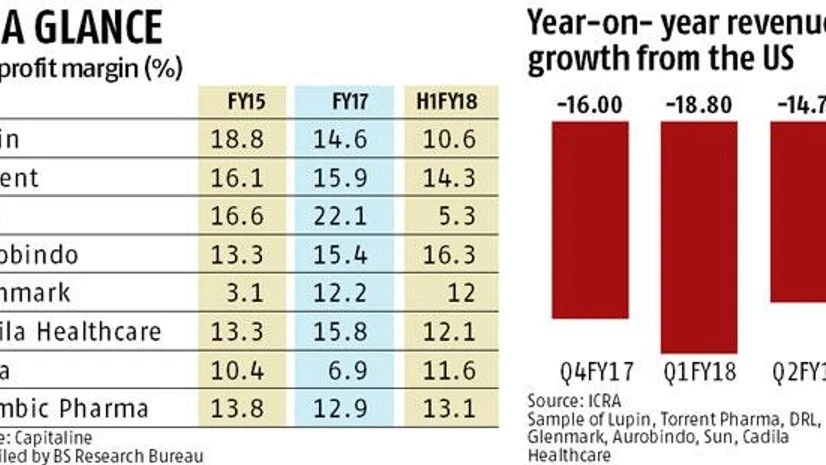
The pace of US Food and Drug Administration (USFDA) approvals has picked up, but that has not necessarily converted into better profit margins for 'big pharma' in India. In fact, most big pharma firms have seen their margins shrink over the last three years barring a few such as Glenmark, which have seen a boost due to limited competition products.
Indian pharma firms including their subsidiaries have received around 304 abbreviated new drug application (ANDA) approvals from the USFDA in 2017 (compared to 201 ANDA approvals in 2016). While this is expected to translate into higher revenues in the mid to long-term future, rising competition in the US generic market has affected the margin growth for big pharma.
Analysts expect margins to remain under stress for the coming quarters as first to file (FTF) opportunities are limited. FTF basically imply that the company getting the nod would enjoy a 180-day exclusivity period for selling that drug.
Gaurav Jain, vice president, corporate ratings, Icra Limited pointed out that the operating margins of the major pharma companies fell from 26-27 per cent in Q4FY17 to 18-19 per cent in the last three quarters or so despite cost optimisation measures undertaken by these companies. In the Icra sample (Sun, Lupin, Torrent, Cadila, DRL, Glenmark, Aurobindo) the revenue growth from the US has been negative for the past few quarters; it fell by 16 per cent year-on-year (y-o-y) in Q4FY17, then again fell by 18.8 per cent in Q1FY18 and could not recover in Q2FY18 either (-14.7 per cent).
Jain feels that apart from price erosion, the FTF opportunities too have been limited (Cadila Healthcare and Aurobindo having one each in last few quarters). Price erosion has been primarily contributed by consolidation in US distribution supply chain in which three large buying consortiums control around 85 per cent of the drug purchase and also the faster pace of ANDA approvals. As such the pace of USFDA approvals has been on the rise - from 440 ANDA approvals in October 2012 to September 2013 period, it rose to 763 in October 2016 to September 2017 period.
Companies like Lupin saw its net profit margins shrink from 18.8 per cent in FY15 to 14.6 per cent in FY17 and 10.6 per cent in H1FY18; for Torrent, it has shrunk from 16.1 per cent to 14.3 per cent for the same period.
Glenmark has seen its margins improve over a two-year horizon though - from 3 per cent in FY15 to 12 per cent in FY17 due to the launch of generic anti-cholesterol drug Zetia (180-day marketing exclusivity) in the US in December 2016. The drug has contributed around $60 million or over a third of the company's US generic sales in the third quarter of FY17. As the six month's exclusivity period ended in the June quarter of FY18, the company has started witnessing slower growth in revenues (1.5 per cent) in Q2FY18 and a decline in operating and net profits (down by 13.5 per cent and 4.2 per cent respectively in Q2FY18 compared to the year before period).
As for India's most valuable pharma company Sun Pharma, pricing pressures have eaten into its US business - in Q2FY18, Sun's revenues from its cancer drug Gleevec were down by 40 per cent on a sequential basis due to competition from Teva and Apotex. In FY17 this drug contributed to $230 million of Sun's sales.
On the whole, analysts feel that the US business would continue to see pricing pressure in the near to medium term and while revenues might get a boost thanks to higher drug launches, the margins may remain stretched.

)