Clinical trial begins to assess plasma efficacy to limit Covid-19: ICMR
Convalescent plasma is one of several emerging therapies. However, there is no robust evidence to support it for routine therapy
)
premium
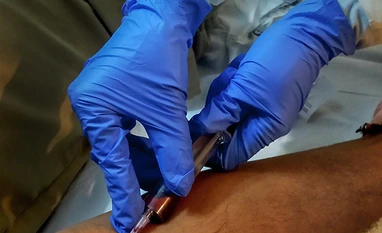
A medic provides medicine as part of Covid-19 treatment at Sri H. N. Reliance Foundation Hospital in collaboration with the Brihanmumbai Municipal Corporation (BMC), in Mumbai. Photo: PTI
The Indian Council of Medical Research has started a multi-centre clinical trial to assess the safety of the convalescent plasma to limit any coronavirus related complications.
Topics : Coronavirus
