Sun, Ranbaxy may be asked to sell some businesses for CCI approval to merger
)
Ranbaxy Laboratories and Sun Pharmaceutical Industries are likely to be advised to sell parts of their businesses before the Competition Commission of India (CCI) approves their merger.
A source said the competition watchdog would by next month suggest "structural remedies" that included selling key drug segments as conditions for clearing the country's largest pharmaceutical industry merger.
When contacted, both Sun Pharma and Ranbaxy refused to comment.
On April 6, Sun Pharma announced it would buy Ranbaxy from Japan's Daiichi Sankyo in a $4-billion all-share deal. Sun Pharma had then said it expected to close the deal by December.
CCI can take up to 210 days to clear a merger. After that a proposed deal is deemed to have been approved.
"Around five months have gone down since the companies applied for approval. We have written to the companies and sought their responses. They have said they can make changes. We need to close the issue by November-end," a CCI official said.
The competition watchdog is writing to Ranbaxy and Sun Pharma suggesting changes to get the deal going. The regulator will also set up "a monitoring agency to ensure adherence consequent to the order", according to the source.
The regulator had sought comments from the public and stakeholders on the merger following concerns that the presence of both companies in key drug segments could lead to monopoly pricing. Consumer forums have argued the merger could stifle competition.
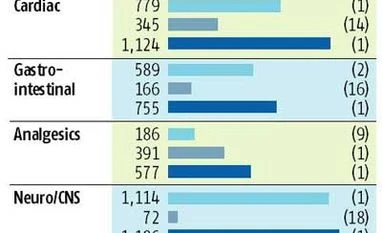
Industry analysts said there was likely to be significant overlap in Ranbaxy's and Sun Pharma's anti-infective and gastro-intestinal drug businesses. Market shares would also be affected in other therapeutic segments like cardiology, analgesics, respiratory, neurology, the central nervous system and gynaecology.
Sun Pharma-Ranbaxy's combined annual revenue is estimated at $4.2 billion - $1.1 billion of that from sales in India. Once the deal is done, Sun Pharma will be the largest drug maker in the country, with a market share of 9.2 per cent. The merger will create the fifth-largest generic drug company in the world.
Sun Pharma will also acquire Ranbaxy's assets in India and in other countries. These include Ranbaxy's factories at Paonta Sahib (Himachal Pradesh), Dewas (Madhya Pradesh), Mohali and Toansa (Punjab) that were earlier supplying drugs to the US.
The CCI approval will allow Sun Pharma to move ahead with its integration plan. Among the issues it will have to address is attrition at Ranbaxy. The Gurgaon-headquartered company's India office has seen several recent departures. Last week, at least five Ranbaxy executives in the US, including country head Venkatachalam Krishnan and heads of the sales, finance and legal departments, resigned together.
On Tuesday, Sun Pharma shares rose 0.09 per cent from their previous close on BSE to end at Rs 804.30. Ranbaxy's shares declined 0.03 per cent to Rs 598.05.
More From This Section
Don't miss the most important news and views of the day. Get them on our Telegram channel
First Published: Oct 15 2014 | 12:57 AM IST
